Informing surgical decisions
with validated biomechanics
CREDIBLE | SIMPLE | EFFICIENT
CLINICALLY GROUNDED
Published retrospective foundational evidence
3D
From Standard X-Rays No mandatory CT-Scan required
30+
Years of Biomechanics Research Arts & Métiers / LBM lineage
V&V40
ASME Standard Compliant biomechanical model verification & validation
WHAT WE DO
Biomechanical intelligence, pre-operatively
Today, surgical decisions for complex spine cases rely on geometric planning and clinical experience. SKAIROS adds a missing layer: the mechanical behavior of the implant, inside this specific patient’s spine.
Patient-specific simulation
3D spine reconstruction from standard frontal-lateral X-rays. No mandatory CT-Scan. Each simulation reflects the patient’s anatomy, alignment, and bone morphology.
Implant behavior comparison
Multiple surgical scenarios are simulated simultaneously. Mechanical load levels at instrumented and adjacent levels are compared across construct strategies. Results are provided as comparative indicators to support surgical planning, not as absolute predictive values.
Scientific rigor, clinical workflow
FEM models validated in vitro, compliant with ASME V&V40. The service integrates into existing pre-operative workflows without additional imaging or procedural changes.

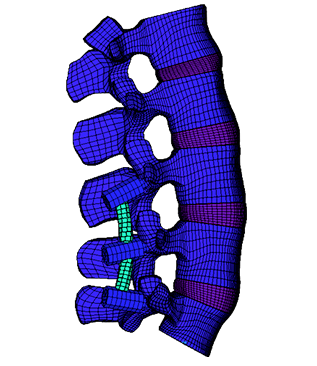
Example of kinematic behavior of an instrumented lumbar segment
Range of motion deliberately accentuated for the demo.
Courtesy of Dr. P. Fayada – Surgeon
TECHNOLOGY
Three uniquely combined properties
CREDIBLE
Highest Validation Standards
FEM models verified in vitro against normative mechanical testing. Compliant with ASME V&V40 — the reference standard for medical simulation credibility. Validated on implant mechanical behavior, anatomical variability, and instrumented spine dynamics - not a generic simulation engine.
SIMPLE
Zero workflow disruption
Full patient-specific 3D spine reconstruction from standard frontal-lateral X-rays alone. No mandatory CT-Scan required. No additional imaging. No change to pre-operative protocols. Operates on what every surgeon already has.
EFFICIENT
Enabling informed surgical strategies
Retrospective evidence validated over 20 years. Now extended to long constructs and adult deformity, with a prospective pivotal study planned.
WHO WE WORK WITH
Two interconnected pathways
Spine Surgeons
For complex cases – multi-level fusions, adult deformity, and innovative non-fusion approaches – where standard planning reaches its limits. SKAIROS provides biomechanical information that complements clinical judgment on complex cases.
-
Pre-operative scenario comparison
-
Mechanical stress comparisons across surgical strategies
-
Implant selection support
-
Topping-off strategy evaluation
-
Can be leveraged in training / educational contexts
Implant Manufacturers
Note: SKAIROS is currently in Research-use phase, targeting CE / FDA mark. Regulatory-grade documentation is produced in anticipation of clinical deployment.
From pre-market validation to clinical evidence generation. SKAIROS integrates your implant into a validated biomechanical platform – producing scientific data, surgeon tools, and regulatory-grade documentation.
-
Implant FEM modeling & in vitro V&V
-
Integration into SKAIROS surgical planning software
-
Retrospective clinical data analysis
-
Support to regulatory dossier (CE / FDA)
SCIENTIFIC FOUNDATION
Built on decades of Clinical Biomechanics Research
1 • Finite Element Models
Spine-specific FEM covering intact, injured, and instrumented configurations. Validated against in vitro experimental data across multiple spinal regions.
2 • Quantitative Image Analysis
Validated tools for extracting clinical and biomechanical parameters from biplanar X-rays feeding directly into the simulation pipeline.
3 • ASME V&V40 Compliance
All models follow the ASME V&V40 standard for credibility assessment of computational models applied to medical devices.
4 • SaMD Regulatory Pathway
Currently in Research-use, SKAIROS’ Solution is developed as a Software as a Medical Device (SaMD), targeting CE Mark (EU MDR) and FDA clearance. The platform architecture is designed for regulatory scalability.
OUR TEAM
Biomechanics & clinical expertise at its core

Wafa Skalli
Co-Founder, CEO & CSO
Eng. & PhD ENSAM, Honorary Professor. Clinical biomechanics & numerical simulation.

Aurélien Laville
COO
Eng. & PhD ENSAM. 15 years expertise in 3D technologies & medical imaging.

Alexandre Templier
VP Strat. Dev.
Eng., PhD & MBA. >25 years expertise in AI-enabled decision support.

Ali Benkirane
CFO
Economic, financial & social analysis.

Virginie Lafage
Scientific Advisor
Eng. & PhD ENSAM, Lenox Hill Hospital NY. Clinical biomechanics, multicentric studies.

Guy Viart
Board Advisor
Industry expertise in spinal implants & medtech commercialization.
OUR PARTNERS
A trusted ecosystem
INSTITUTIONAL





ACADEMIC




CLINICAL





THEY TRUST SKAIROS
What our partners say
NEWS
Latest News from SKAIROS
CONTACT US
Let’s discuss your needs
Whether you are a spine surgeon looking to explore biomechanical planning, or an implant manufacturer seeking to integrate your device into a validated simulation platform – we’d be glad to hear from you.
-
ADRESS: Pépinière Paris Santé Cochin, 29 rue du Faubourg Saint-Jacques – 75014 PARIS
-
EMAIL: contact@skairos.io
-
LINKEDIN: linkedin.com/company/skairos





